Table of Contents
- 1 Step-By-Step Process of Supplement Registration
- 2 What Is Food Supplement Registration in Nepal?
- 3 Where To Register Food Supplements in Nepal?
- 4 How To Register Food Supplements in Nepal?
- 5 What Documents Are Required for Food Supplement Registration?
- 6 How Long Does Food Supplement Registration Take in Nepal?
- 7 What Are the Costs for Food Supplement Registration?
- 8 What Are Post-Registration Requirements for Food Supplements?
- 9 What Types of Food Supplements Require Registration in Nepal?
- 10 What is the Process of Renewing Food Supplement in Nepal?
- 11 What is the Process for Import of Food Supplement in Nepal?
- 12 What Benefits Does Food Supplement Registration ?
- 13 What Laws Govern Food Supplement Registration in Nepal?
- 14 Frequently Asked Questions
- 14.1 What is food supplement registration in Nepal?
- 14.2 How long does food supplement registration take?
- 14.3 What documents are needed for registration?
- 14.4 Can I import supplements without registration?
- 14.5 What are the heavy metal limits in Supplements?
- 14.6 How much does registration cost?
- 14.7 What information must Supplement Label contain?
- 14.8 How to Renew Supplement in Nepal?
- 14.9 How to register a food company in Nepal?
Food supplement registration in Nepal is a mandatory regulatory process administered by the Department of Food Technology and Quality Control (DFTQC). Registration requires mandatory laboratory tests, HACCP certification, and registration in the home country.
The process takes 40–120 working days. This guide provides information on the laws, required documents, registration process, timelines, costs, import procedures, renewal procedures, and limitations related to food supplements in Nepal.
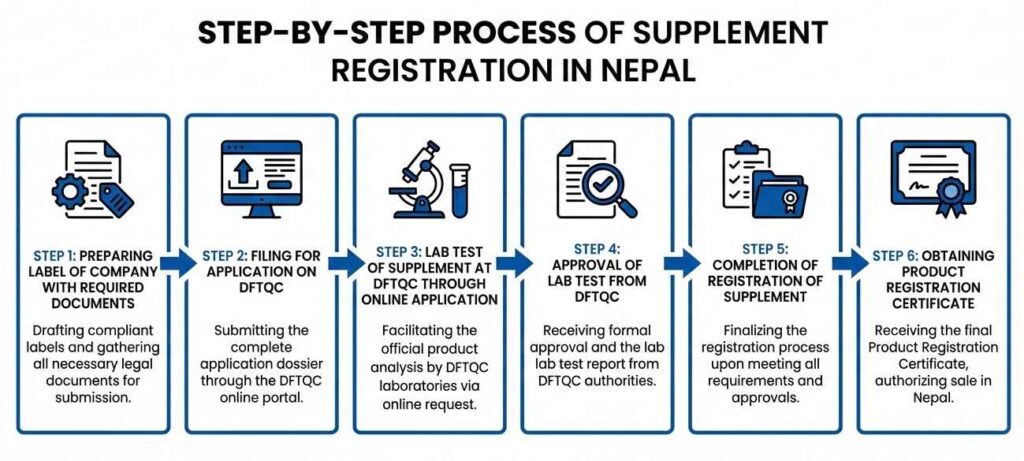
Step-By-Step Process of Supplement Registration
- Step 1: Preparing Label of Company with Required Documents
- Step 2: Filing for Application on DFTQC
- Step 3: Lab Test of Supplement at DFTQC through Online Application
- Step 4: Approval of Lab Test from DFTQC
- Step 5: Completion of Registration of Supplement
- Step 6: Obtaining Product Registration Certificate
What Is Food Supplement Registration in Nepal?
Food supplement registration in Nepal is the official authorization process required for dietary supplements, nutraceuticals, health supplements, and special dietary purpose foods before production, import, sale, or distribution. The Department of Food Technology and Quality Control oversees this registration under the Dietary Supplement Directives, 2072. This applies to various categories including foods for special dietary purposes, foods for special therapeutic use, nutraceuticals, and nutritional supplements.
Food supplement registration in Nepal requires coordination between importers holding valid import-export licenses and manufacturers possessing ISO 22000 certification, GMP certification, and free sale certificates. Products must be registered in their country of origin and pass laboratory testing according to Nepal’s guidelines.
Where To Register Food Supplements in Nepal?
Food supplement registration in Nepal must be completed at the Department of Food Technology and Quality Control (DFTQC), the primary regulatory authority under the Ministry of Agricultural Development responsible for food safety, quality control, and product certification. The DFTQC headquarters located in Kathmandu processes all registration applications, conducts laboratory testing, and issues product registration certificates for dietary supplements and nutraceuticals.
Applicants submit registration documents through the DFTQC online portal system, which facilitates application tracking, document submission, and communication with regulatory officials. The DFTQC laboratory facilities conduct mandatory testing for heavy metals, microbial contamination, active ingredients, and nutritional composition verification.
For detailed information and application procedures, visit the official DFTQC . The Ministry of Agricultural Development provides additional regulatory guidance. Importers must also coordinate with the Department of Customs for import procedures.
How To Register Food Supplements in Nepal?
- Step 1: Preparing Label of Company with Required Documents
- Step 2: Filing for Application on DFTQC
- Step 3: Lab Test of Supplement at DFTQC through Online Application
- Step 4: Approval of Lab Test from DFTQC
- Step 5: Completion of Registration of Supplement
- Step 6: Obtaining Product Registration Certificate
Step 1: Verify Eligibility of Product with Certifications
The distributor must possess valid import-export license registration in Nepal with clearly mentioned capital and permission for dietary supplement import. The manufacturer must hold ISO 22000 certification, GMP certification, and free sale certificate from the country of origin. Verify product registration in the parent country and ensure compliance with Nepal’s quality standards.
Step 2: Prepare Product Label and Documentation
Design product label containing product name, identity (dietary supplement/health supplement/nutraceuticals), ingredient composition with RDA percentages, quantity, manufacturer details, manufacturing and expiry dates, batch number, health claims, recommended use, warnings, disclaimer stating “not intended to treat, cure or diagnose any diseases,” and importer details including name, address, EXIM code, and email.
Step 3: Compile Required Documents for Registration
Gather importer documents including application form, firm registration certificate, citizenship certificate, PAN, VAT, EXIM code, and brand name declaration. Collect manufacturer documents including production license, contract manufacturing agreement, warranty letter, free sale certificate, product registration proof, GMP certificate, HACCP or ISO 22000 certificate, colored label design, raw material specifications, product formulation, master formula card, production flowchart, and site master file.
Step 4: Submit Application to DFTQC
File registration application through the DFTQC online portal with complete documentation and product samples. Pay registration fees as per license issuance fee under Rule 28 of Food Regulations. Submit valid certificate of analysis from in-house and third-party government-approved laboratories showing active ingredient tests, contaminant tests for microbiological parameters, heavy metals, food additives, and residues.
Step 5: Laboratory Testing at DFTQC
Submit product samples for mandatory laboratory testing at DFTQC facilities. The department conducts analysis for heavy metals (cadmium, arsenic, lead, mercury), microbial contamination (yeast, mold, salmonella, E. coli, staphylococcus aureus), active ingredients verification, and nutritional composition. Testing ensures compliance with maximum permissible limits: cadmium not exceeding 1.5 ppm, arsenic 1.1 ppm, lead 2.5 ppm, mercury 1 ppm, with microbial pathogens absent.
Step 6: Receive Product Registration Certificate
Upon successful laboratory test approval and documentation verification, DFTQC issues the product registration certificate in prescribed format. The certificate authorizes legal production, import, sale, and distribution of the registered food supplement in Nepal. Maintain certificate validity through timely renewal as per Rule 31 of Food Regulations regarding license duration and renewal procedures.
What Documents Are Required for Food Supplement Registration?
Importer’s Documents:
- Application form duly filled, signed, and stamped by proprietor
- Firm registration or company certificate with total capital and import/export permission for dietary supplements
- Citizenship certificate, PAN, VAT, and EXIM code certificate
- Declaration letter about brand name
Manufacturer’s Documents:
- Manufacturing license from government authority (DFTQC for Nepali products, FSSAI for Indian products) with valid date
- Legal document for contract manufacturing or authorization letter for marketing/distribution in Nepal (valid and notarized)
- Letter of warranty from manufacturer (valid and notarized)
- Free sale certificate issued by government authority of producing country (for imports)
- Product registration document from government authority of origin country
- Valid and notarized Good Manufacturing Practices (GMP) certificate
- HACCP or ISO 22000 certificate for special dietary, therapeutic, or nutritional purpose foods
- Colored label design
- Raw material specification documents
- Product formulation and scientific rationale
- Master formula card or batch manufacturing record
- Detailed production flow chart
- Site master file of manufacturing unit with sequential photographs
Laboratory Testing Documents:
- Valid and notarized or original certificate of analysis (COA) from in-house and third-party government-approved or accredited laboratory with reference methods
- Active ingredients test results verifying composition claimed on label
- Contaminants test results for microbiological parameters, heavy metals, food additives, and residues
- DFTQC laboratory test report as per requirements
- Test reports matching label claims
Additional Documents:
- Functional properties documentation if applicable
- Dosage specifications and scientific justification
- Toxicity level information
- Molecular warnings for specific population groups
- Molecule free-sale certification confirming non-banned status
Permissible Limit on Food Supplements in Nepal

Under Dietary Supplement Directives, 2072, products must meet specified limits for heavy metal contamination and remain free from microbial pathogens.
| Parameter | Maximum Permissible Limit / Requirement | Testing Requirement |
|---|---|---|
| Cadmium | Not more than 1.5 ppm (mg/kg) | Mandatory heavy metal analysis |
| Arsenic | Not more than 1.1 ppm (mg/kg) | Mandatory heavy metal analysis |
| Lead | Not more than 2.5 ppm (mg/kg) | Mandatory heavy metal analysis |
| Mercury | Not more than 1 ppm (mg/kg) | Mandatory heavy metal analysis |
| Yeast and Mold Count | Should be Absent | Mandatory microbiological testing |
| Salmonella | Should be Absent | Mandatory microbiological testing |
| Escherichia Coli | Should be Absent | Mandatory microbiological testing |
| Staphylococcus aureus | Should be Absent | Mandatory microbiological testing |
| Active Ingredients | As per label claim | Verification against declared composition |
| Nutritional Composition | Minimum 15% of RDA per serving | Nutrient analysis with reference methods |
How Long Does Food Supplement Registration Take in Nepal?
Food supplement registration in Nepal involves stages Submission of Application, Testing of Laboratory and Final Certificate of Issuance around 40-120 Working Days. The registration process typically requires the following timeframes:
- Application Submission and Initial Review: 7-10 working days for DFTQC to review submitted documents and verify application of supplements.
- Laboratory Testing Period: 30-90 days for analysis of heavy metals, microbial contamination, active ingredients, and nutritional composition at DFTQC facilities.
- Certificate Issuance: 5-7 working days after successful approval for DFTQC to prepare and issue the product registration certificate in prescribed format.
- Total Registration Duration: 40-120 working days from complete application submission to certificate issuance, assuming no deficiencies or additional testing requirements.
What Are the Costs for Food Supplement Registration?
Food supplement registration in Nepal involves various fees payable to DFTQC and associated costs for documentation, testing, and compliance verification.
| Fee Category | Amount | Description |
|---|---|---|
| Product Registration Fee | As per Rule 28 of Food Regulations | Charged for each product registration; domestic production exempt from registration fee |
| Laboratory Testing Fee | Variable based on test parameters | Covers heavy metal analysis, microbial testing, active ingredient verification, and nutritional composition analysis |
| Customs Duty for Import | As per Import Export Inspection Directive | Charged for import approval application processing before product importation |
| Document Notarization Costs | Depends | For notarization of manufacturer agreements, warranty letters, GMP certificates, and authorization documents |
| Renewal Fee | As per Rule 31 of Food Regulations | Charged for registration certificate renewal before expiry period |
| Customs Clearance Fee | Variable based on product value | Import duties, taxes, and inspection fees at customs points |
What Are Post-Registration Requirements for Food Supplements?
Food supplement registration in Nepal requires ongoing compliance with regulatory standards, monitoring obligations, and periodic renewals to maintain legal market authorization.
- Registration Renewal: Submit renewal application as per Rule 31 of Food Regulations before certificate expiry with updated documentation, compliance verification, and renewal fees.
- Label Compliance Maintenance: Ensure all product labels continue meeting requirements under Rule 18 of Food Regulations and Clause 18 of Dietary Supplement Directives, 2072, including accurate ingredient declarations, health claims, warnings, and importer details.
- Import Clearance for Each Shipment: Obtain examination and clearance from DFTQC or subordinate offices at customs points for every imported consignment before market release.
- Manufacturing Practice Compliance: Continue applying Good Manufacturing Practices (GMP), ISO 22000, or HACCP standards in production facilities with periodic verification by regulatory authorities.
- Label Modification Approval: Obtain DFTQC approval before implementing any changes to product formulation, label design, health claims, or ingredient composition.
- Market Surveillance Compliance: Ensure products in market circulation maintain quality standards and do not exceed expiry dates as per Clause 24 of Dietary Supplement Directives, 2072.
What Types of Food Supplements Require Registration in Nepal?
Food supplement registration in Nepal applies to various categories of dietary products, nutraceuticals, and special purpose foods as defined under Dietary Supplement Directives, 2072.
| Supplement Type | Description | Requirement |
|---|---|---|
| Dietary Supplements | Products containing vitamins, minerals, amino acids, enzymes, or other nutritional substances intended to supplement normal diet | Mandatory registration before production, import, or sale |
| Health Supplements | Nutritional products designed to support general health, wellness, and physiological functions without therapeutic claims | Mandatory registration with scientific rationale and formulation details |
| Nutraceuticals | Food products providing health benefits beyond basic nutrition, including disease prevention or treatment support | Mandatory registration with clinical evidence and safety documentation |
| Foods for Special Dietary Purposes | Products formulated for specific dietary requirements due to physical condition, disease, or nutritional needs | Mandatory registration with HACCP or ISO 22000 certification |
| Foods for Special Therapeutic Use | Nutritional products designed for specific medical conditions requiring dietary management under professional supervision | Mandatory registration with therapeutic justification and medical evidence |
| Foods for Special Nutritional Purpose | Products addressing particular nutritional deficiencies or supporting specific physiological conditions through targeted nutrient delivery | Mandatory registration with nutritional composition verification |
| Protein Supplements | Products containing concentrated protein sources including whey, casein, soy, or plant-based proteins for muscle support | Mandatory registration with protein content verification and purity testing |
| Vitamin and Mineral Supplements | Products providing concentrated vitamins, minerals, or trace elements to address nutritional gaps or deficiencies | Mandatory registration with RDA compliance and dosage verification |
What is the Process of Renewing Food Supplement in Nepal?
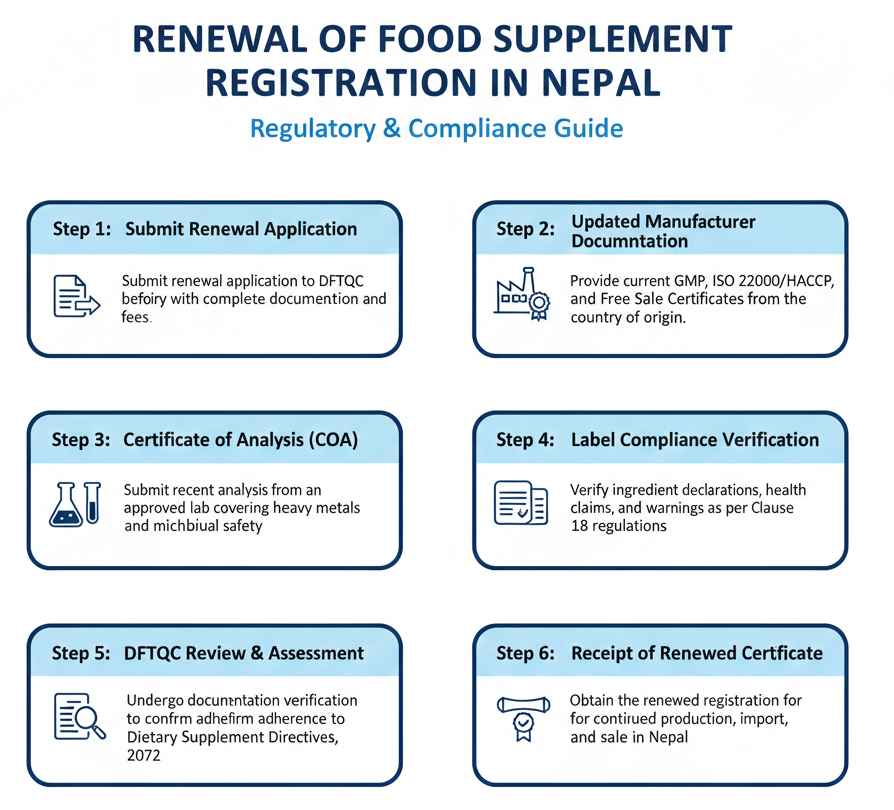
Food supplement registration renewal in Nepal is conducted as per Rule 31 of Food Regulations:
- Step 1: Submit renewal application to DFTQC before current registration certificate expiry date using prescribed format with complete documentation and renewal fees.
- Step 2: Provide updated manufacturer documents including current GMP certificate, ISO 22000 or HACCP certification, free sale certificate, and product registration proof from country of origin.
- Step 3: Submit recent certificate of analysis from government-approved laboratory showing compliance with heavy metal limits, microbial safety standards, and nutritional composition specifications.
- Step 4: Verify label compliance with current regulations including accurate ingredient declarations, health claims, warnings, and importer contact information meeting Clause 18 requirements.
- Step 5: Undergo DFTQC review of renewal application, documentation verification, and compliance assessment to confirm continued adherence to Dietary Supplement Directives, 2072.
- Step 6: Receive renewed product registration certificate upon successful approval, maintaining legal authorization for continued production, import, sale, and distribution in Nepal.
What are the Required Documents for Renewal?
- Renewal application form duly completed and signed
- Current product registration certificate copy
- Updated firm registration and EXIM code certificate
- Valid manufacturer license and GMP certification
- Recent certificate of analysis from accredited laboratory
- Updated free sale certificate from country of origin
- Current product label design and formulation details
- Renewal fee payment receipt as per regulations
- Declaration of no formulation or label changes (if applicable)
- Authorization letter from manufacturer for continued distribution
What is the Process for Import of Food Supplement in Nepal?
Food supplement import in Nepal requires compliance with Dietary Supplement Directives, 2072, and Import Export Inspection and Quality Certification Directive, 2063.
- Step 1: Obtain valid import-export license with specific permission for dietary supplement import from Department of Industry or relevant authority with clearly mentioned capital requirements.
- Step 2: Secure product registration certificate from DFTQC before initiating import procedures, ensuring supplement meets Nepal’s quality standards and regulatory requirements under Clause 13.
- Step 3: Apply for import approval from DFTQC as per Sub-clause (a) of Clause 11 and Clause 15, submitting manufacturer documents, product formulation, laboratory analysis, free sale certificate, and authorization letter.
- Step 4: Submit product samples for DFTQC analysis if required under Clause 16, paying analysis fees and awaiting laboratory testing results confirming compliance with safety parameters.
- Step 5: Coordinate with customs authorities at entry points, presenting import approval, product registration certificate, commercial invoice, packing list, and certificate of analysis for clearance procedures.
- Step 6: Undergo mandatory examination and clearance by DFTQC or subordinate offices at customs points as per Sub-clause (b) of Clause 11 before releasing products for market distribution.
- Step 7: Ensure imported products maintain proper storage conditions, valid expiry dates, and label compliance throughout distribution chain as per Clause 24 and labeling requirements.
Import Requirements Under Dietary Supplement Directives, 2072:
- Only official sellers of foreign producers may import food supplements as per Sub-clause (c) of Clause 11
- Manufacturer must possess production license from food hygiene regulatory authority of origin country
- Valid and notarized contract manufacturing agreement or authorization letter for Nepal distribution required
- Free sale certificate from government authority confirming no sales restrictions in origin country mandatory
- Product must be registered in government authority of producing country with supporting documentation
- GMP certification demonstrating recognized manufacturing practices application in production process essential
- Laboratory testing from accredited facilities showing nutrient analysis and hygiene standards compliance necessary
- Import approval must be obtained before shipment arrival at customs points for regulatory clearance
- Each imported consignment requires examination and clearance by DFTQC before market release
- Imported products must comply with heavy metal limits, microbial safety standards, and labeling requirements
For import procedures and regulations, visit the Department of Customs and Ministry of Industry, Commerce and Supplies.
READ MORE: Obtaining DFTQC License in Nepal
What Benefits Does Food Supplement Registration ?
Food supplement registration in Nepal offers significant advantages for manufacturers, importers, consumers, and the regulatory system by ensuring product safety, market legitimacy, and consumer protection.
- Registration provides official permission for production, import, sale, and distribution of food supplements throughout Nepal.
- Registered products undergo rigorous laboratory testing for heavy metals, microbial contamination, and nutritional composition.
- Registration process validates product formulation, manufacturing practices, and ingredient accuracy.
- Registered products receive streamlined customs clearance at entry points.
- Registration ensures adherence to Dietary Supplement Directives, 2072, Food Act, Food Regulations, and international standards.
What Laws Govern Food Supplement Registration in Nepal?
Food supplement registration in Nepal operates under these legal frameworks. The following laws and directives regulate dietary supplements, nutraceuticals, and special dietary purpose foods:
- Dietary Supplement Directives, 2072: Primary regulatory framework governing production, sale, distribution, import, and export of food supplements, nutraceuticals, foods for special dietary purposes, and foods for special therapeutic purposes in Nepal.
- Food Act, 2023 (1966): Foundational legislation establishing food safety standards, quality control measures, licensing requirements, and penalties for food-related violations.
- Food Regulations, 2027 (1970): Implementing regulations under the Food Act detailing licensing procedures, registration fees, renewal processes, labeling requirements, and quality standards for food products.
- Import Export Inspection and Quality Certification Directive, 2063: Governs import approval procedures, customs clearance, quality inspection, and certification requirements for imported food supplements and dietary products.
- Codex Alimentarius Commission Standards: International food quality standards adopted by Nepal for nutrients, food additives, preservatives, and hygiene standards not specifically covered under national regulations assisted by law firm in nepal.
- Consumer Protection Act, 2075 (2018): Provides consumer rights protection, prohibits misleading claims, and establishes penalties for false advertising or substandard food supplement products.
Frequently Asked Questions
What is food supplement registration in Nepal?
Food supplement registration in Nepal is the mandatory regulatory approval process administered by DFTQC for dietary supplements, nutraceuticals, and special dietary purpose foods. Registration ensures products meet safety standards, quality parameters, and labeling requirements before legal production, import, sale, or distribution in Nepal’s market.
How long does food supplement registration take?
Step 1: Submit complete application with documents (7-10 days review)
Step 2: Laboratory testing at DFTQC facilities (15-30 days)
Step 3: Technical evaluation and compliance assessment (10-15 days)
Step 4: Certificate preparation and issuance (5-7 days)
Step 5: Total duration approximately 40-60 working days
What documents are needed for registration?
The Documents for Supplement Registration are:
1. Firm registration and EXIM code certificate
2. Manufacturer GMP and ISO 22000 certification
3. Free sale certificate from origin country
4. Product formulation and scientific rationale
5. Certificate of analysis from accredited laboratory
Can I import supplements without registration?
No, food supplement import without DFTQC registration is prohibited under Clause 3 of Dietary Supplement Directives, 2072. Products must obtain registration certificate before importation, and each shipment requires examination and clearance by DFTQC or subordinate offices at customs points for legal market entry.
What are the heavy metal limits in Supplements?
Cadmium maximum 1.5 ppm, arsenic maximum 1.1 ppm, lead maximum 2.5 ppm, mercury maximum 1 ppm. Microbial pathogens including yeast, mold, salmonella, E. coli, and staphylococcus aureus must be absent. Laboratory testing verifies compliance before registration approval and market authorization.
How much does registration cost?
Step 1: Product registration fee as per Rule 28
Step 2: Laboratory testing fee (variable by parameters)
Step 3: Sample analysis fee paid by applicant
Step 4: Document notarization costs (NPR 500-2,000)
Step 5: Third-party COA costs (NPR 5,000-15,000)
What information must Supplement Label contain?
Product name, identity (dietary supplement/nutraceuticals), ingredient composition with RDA percentages, quantity and dosage, manufacturer details, manufacturing and expiry dates, batch number, health claims, recommended use, warnings, disclaimer stating “not for medicinal use,” and complete importer details including name, address, and EXIM code.
How to Renew Supplement in Nepal?
Submit renewal application before certificate expiry with updated manufacturer documents, current GMP certification, recent certificate of analysis, label compliance verification, and renewal fees. DFTQC reviews documentation, verifies continued compliance with Dietary Supplement Directives, 2072, and issues renewed certificate upon successful approval.
How to register a food company in Nepal?
1. Register the business with the Office of Company Registrar.
2. Obtain a PAN/VAT certificate from the Inland Revenue Department.
3. Apply for a food production license from the Department of Food Technology and Quality Control (DFTQC).
4. Secure necessary health, safety, and quality control approvals.




































